1. Clear MoA:
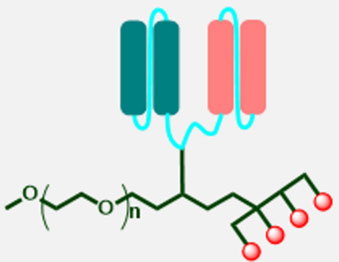
(1) Antibody-drug conjugates (ADCs) are a rapidly growing class of cancer therapeutics that seek to overcome the low therapeutic index of conventional cytotoxic agents. However, realizing this goal has been a significant challenge. ADCs comprise several independently modifiable components, including the antibody, payload, linker, and bioconjugation method. Many approaches have been developed to improve the physical properties, potency, and selectivity of ADCs.
(2) ADC stands for antibody-coupled drug, which is formed by coupling a monoclonal antibody with a small molecule drug. This drug has the characteristics of high specificity and good safety, which can effectively kill tumor cells and be used in the clinical treatment of tumor diseases. The mechanism of action of ADC drugs is mainly through the targeting effect of monoclonal antibodies to specifically recognize the antigens on the surface of tumor cells, and then the endocytosis of the cells in the patient's body can make the chemical drugs enter the tumor cells in the patient to achieve the purpose of killing the tumor cells in the body. Since the small-molecule drugs of ADC drugs are generally released after they enter tumor cells, the safety of ADC drugs can be improved, the side effects of drugs can be reduced, and the effectiveness of drugs can be enhanced.
(3) This molecule is the first reported P-BsADC that introduce bispecific antibody as its component and used PEG as its framework.
2. Excellent molecular design:
(1) Site-specific conjugation to reduce mismatch rate of bispecific antibody and control drug-to-antibody ratios (DAR) precisely, resulting in homogenous and stable end-product. Moreover, simple organic synthesis could scale up easily.
(2) No Fc/FcR mediated non-target toxicity effect, by substituting Fc fragment with PEG.
3. Excellent preclinical result:
(1) Low binding-site barrier due to small molecular weight compared to marketed ADC, high efficiency of endocytosis with low excretion results in high utilization ratio of the drug, make this P-BsADC a promising candidate for the treatment of solid tumor.
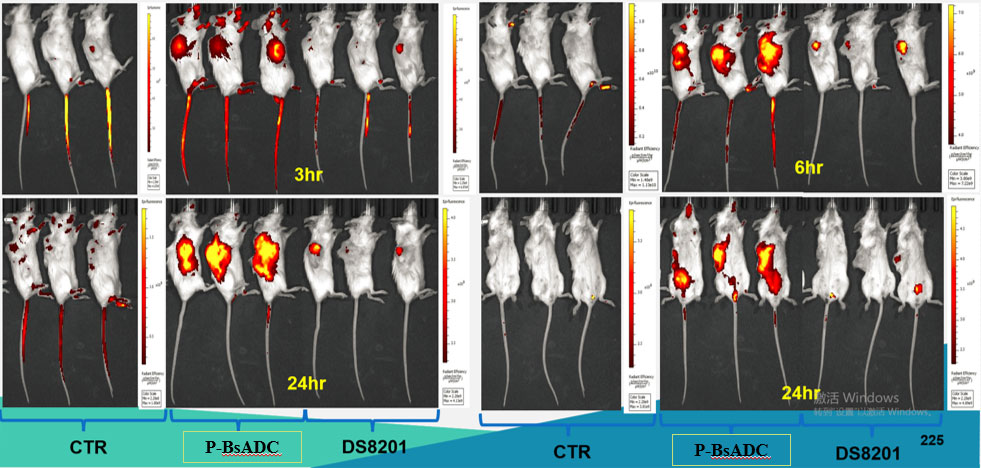
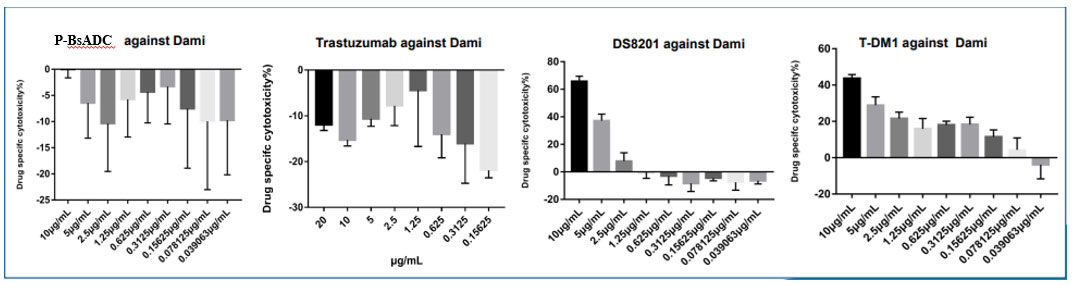
(2) Excellent bystander effect compared with DS8201 and T-DM1 in cells.
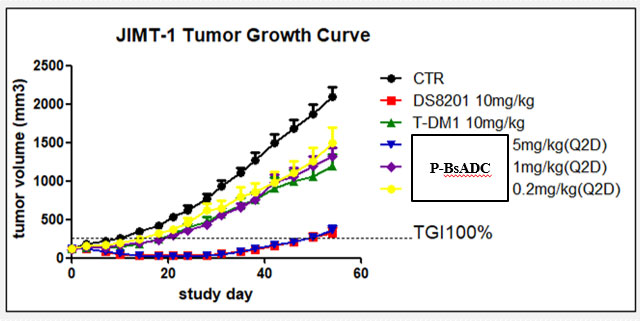
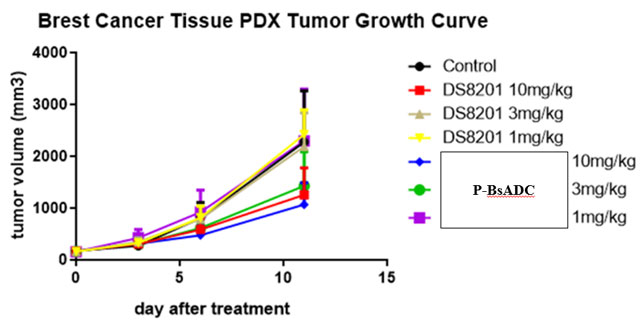
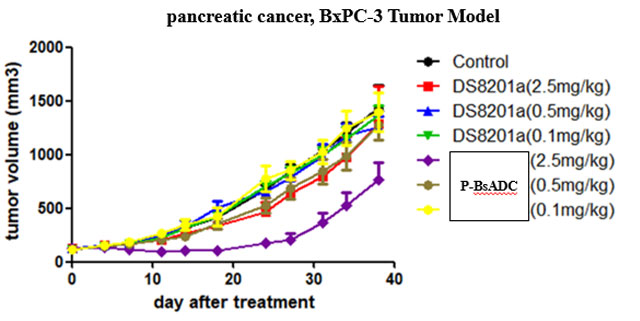
(3) Tumor inhibiting ability was better than T-DM1 and DS8201a in both HER2 high and low expression models at the same dose. (Breast cancer, pancreatic cancer)
(4) Without Fc, no cytotoxicity to megakaryocytes was observed, its huge improvement over DS8201 and T-DM1.
1. Asset type: Anti-Her2 II / Anti-Her2 IV Bi-specific antibody ADC
2. Indication: Solid tumor
3. Research phase: Preclinical
4. Cooperation demands: License-out or co-development
5. Research progress:
(1) Completed proof-of-concept, mechanism research and animal experiments of this project.
(2) In general, this project is superior to DS8201a and T-DM1 in vitro, and is far more effective than DS8201a or T-DM1 in tumor suppression in vivo.
(3) The safety of this project has also been verified to some extent, will carry out further toxicological studies in non-human primates, and expect to obtain better results than DS8201a or T-DM1 in toxicological experiments.
2025.

Explore our catalog of therapeutic antibody solutions to find the right products for you! We are dedicated to delivering solutions designed to help you drive innovation and push the boundaries of what therapeutic antibodies can be.

Organoid Toolbox is a collection of organoid solutions including ready-to-use organoids, organoid differentiation kits, and a variety of services to accelerate the progress of your drug development project.
To enable antibody characterization methodsACROBiosystems has developed a series of enzymes.such as ldeS, SpeB, EndoH, and Endo S proteases, toassist with the characterization of antibodies and theirrelated post-translational modifications (PTMs)

ACROBiosystems developed a series of GMP grade cytokines under the GMP grade quality management system. Those products are all suitable for T/NK cell generation, activation, and proliferation in cell therapy research.

50+ targets designed for CAR detection, including PE/FITC/biotin labeled proteins. The key reagents for CD19 and BCMA were FDA DMF filed which can support your IND, NDA and BLA process.

GMP grade cytokines, reagents for cell activation, gene edition, DNA/RNA removal, etc. Particularly focus on product design, quality control and solution-based support to link each phase of your cell and gene therapy journey.

Full length multi-pass TPs with stabilized structure and high bioactivity for immunization, antibody screening, cell based assay and CAR detection, including hot CD20, Claudin 18.2, CD133, GPRC5D,CCR8, CCR5, etc.

A series of immune checkpoints including classic co-inhibitory and co-stimulatory receptors. The comprehensive catalog contains 100+ targets with various species and tags, and the high-quality proteins are in good batch-to-batch consistency.

To meet the needs of ADCs development, ACROBiosystems can provide: A variety of high-quality target proteins; MMPs/Cathepsin/uPA for cleavable linker; Anti-payload antibodies & anti-idiotypic antibodies for immunogenicity and PK analysis; SPR/BLI analytical and ADA development service.

Comprehensive collection of Fc receptor proteins, including their common variants, which can help expedite your antibody development.

Comprehensive cytokine targets including interleukins, growth factors, chemokines, TNFs, etc. are expressed by HEK293 to ensure their natural structure. Their high purity is verified by SDS-PAGE/HPLC/SEC-MALS and high bioactivity is verified by ELISA/SPR/BLI.

Aneuro provides innovative solutions for neuroscience research. Recombinant proteins, neural factors, pre-formed fibrils, electrophysiological electrodes, as well as Organoid Toolbox all in Aneuro aiming to advance neuroscience research, develop therapeutic interventions, and improve diagnostic methods for neurological diseases.
This web search service is supported by Google Inc.
