 Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit! Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit!
 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
> Multi-pass Transmembrane Proteins Platform 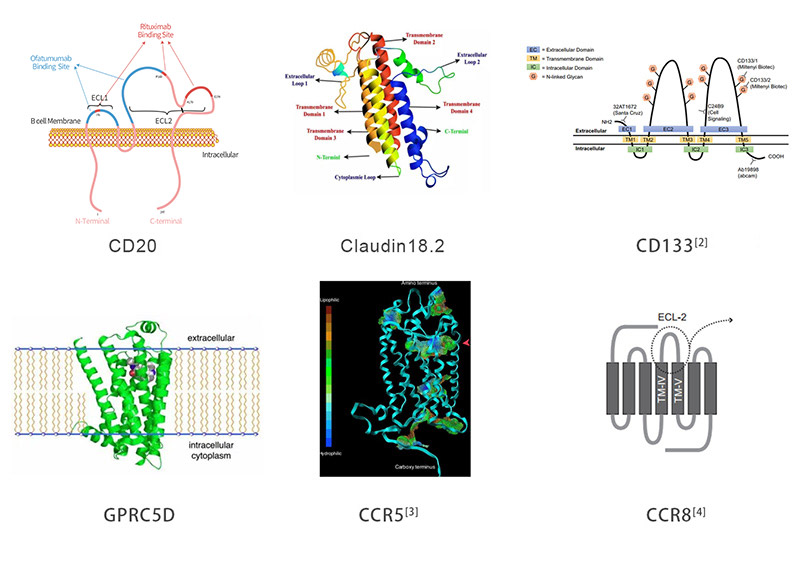
The VLP (Virus like particles) technology platform based on the HEK293 expression system is specially set up by ACROBiosystems to express TPs on the host cell surface. The viral envelop/capsid protein then turns these cell surfaces into soluble lipid bilayer particles with highly-concentrated proteins amenable for antibody immunization and screening. The membrane protein-VLP complex displays correctly folded multi-pass TPs in its cell membrane induce and screen functional antibodies that recognize the natural conformation of the target.In addition to providing VLP based multi-pass TPs in our catalog, ACRO provides customized services as well.
The transmembrane region of multi-pass TPs is highly hydrophobic, and it is difficult to maintain the correct conformation in ordinary buffers once extracted from cell membrane. This problem can be solved by addition of detergents. ACROBiosystems has set up a complete platform for insect cell and mammalian cell expression, purification, and stabilization for difficult drug target TPs. ACRO performs detergent screening including DDM/CHS (Cat. No. DC-11) to increase solubility and ensure native fold of this protein in vitro.
"Nanodisc" is a synthetic phospholipid bilayer membrane structure composed of membrane scaffold proteins (MSPs) and phospholipid molecules. TP can be integrated into the special structure of Nanodisc after removal of the detergent so as to maintains its native folding, retain its biological activity and exhibitimproved hydrophilicity for a wide range of applications. For example, the detergent free formulation of Nanodisc based TP is compatible in CAR expression tests. ACROBiosystems has performed continuous optimization and improvement of the assembly process to make it suitable for industrial scale-up production. ACRO’s efforts have ensured a long-term stable supply of Nanodisc based TP products for the biopharma industry.
VLP
Detergent Micelle
Nanodisc
| Molecule | Cat. No. | Product Description | Application | Preorder/Order |
|---|
| Molecule | Cat. No. | Product Description | Application | Preorder/Order |
|---|
| Molecule | Cat. No. | Product Description | Application | Preorder/Order |
|---|
The following SPR protocols are available for free.
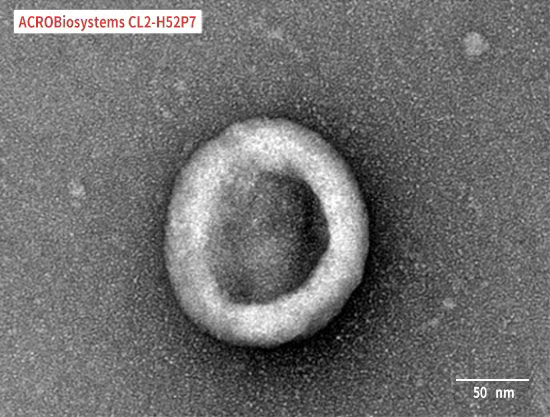

Immobilized Human Claudin-18.2 Full Length Protein-VLP (Cat. No. CL2-H52P7) at 5 μg/mL (100 μL/well) can bind Monoclonal Anti-Chimeric Claudin-18.2 Antibody, Human IgG1 with a linear range of 0.2-3 ng/mL (QC tested).
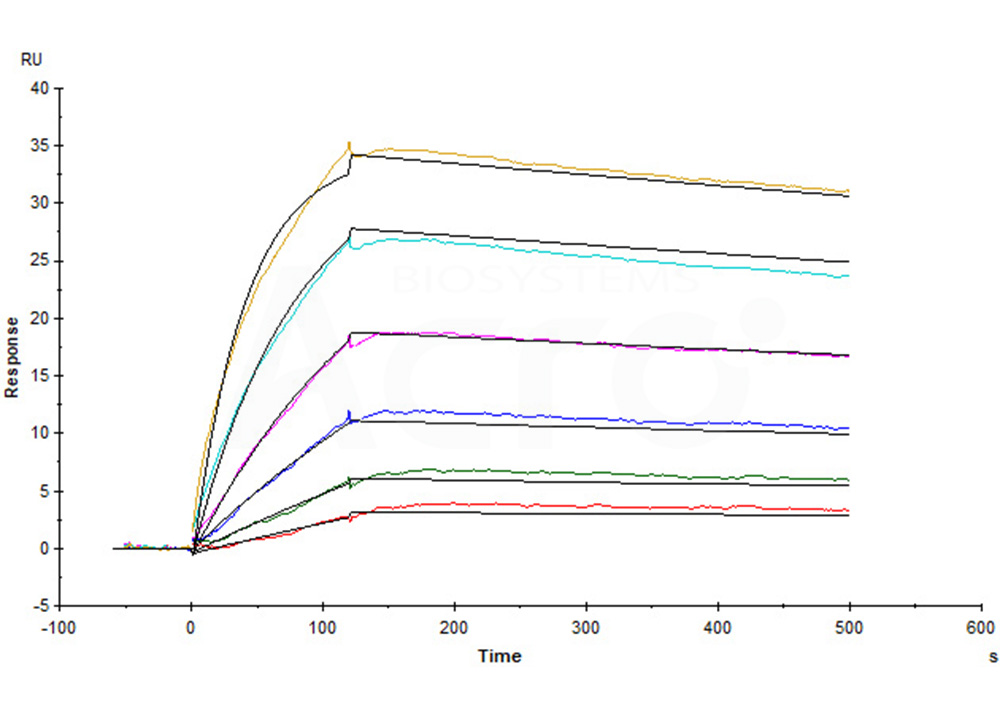
Human Claudin-18.2 Full Length Protein-VLP (Cat. No. CL2-H52P7) captured on CM5 Chip via Anti-Claudin-18.2 antibody can bind Anti-Claudin-18.2 antibody with an affinity constant of 0.374 nM as determined in a SPR assay (Biacore T200) (Routinely tested).
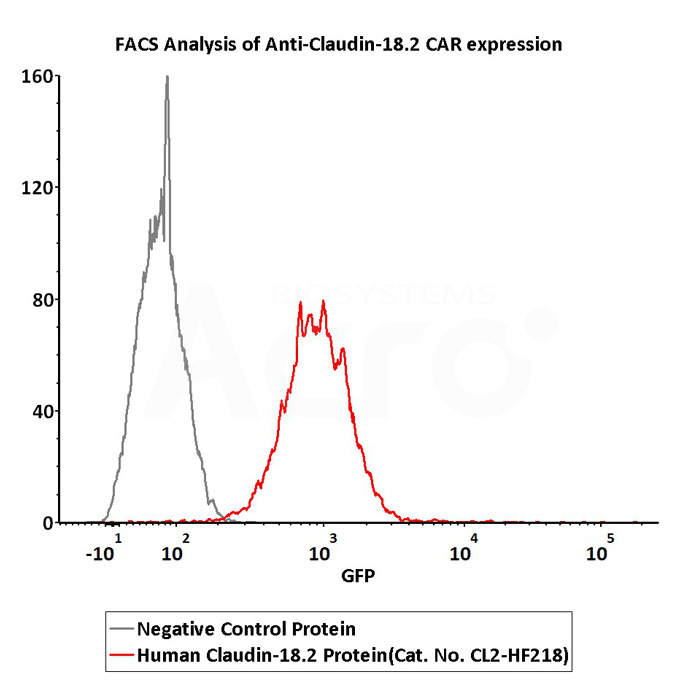
2e5 of Anti-Claudin-18.2 CAR-293 cells were stained with 100 μL of 3 μg/mL of Fluorescent Human Claudin-18.2 Full Length Protein-VLP (Cat. No.CL2-HF218) and negative control protein respectively, FITC signals was used to evaluate the binding activity (QC tested).

Immobilized Rituximab at 2 μg/mL (100 μL/well) can bind Human CD20 Full Length Protein, His Tag (Cat. No. CD0-H52H3) with a linear range of 0.4-3 ng/mL (in presence of DDM and CHS) (QC tested).

Immobilized Rituximab at 2 μg/mL (100 μL/well) can bind Biotinylated Human CD20 Full Length, His,Avitag (Cat. No. CD0-H82E5) with a linear range of 4-63 ng/mL (in presence of DDM and CHS) (QC tested).
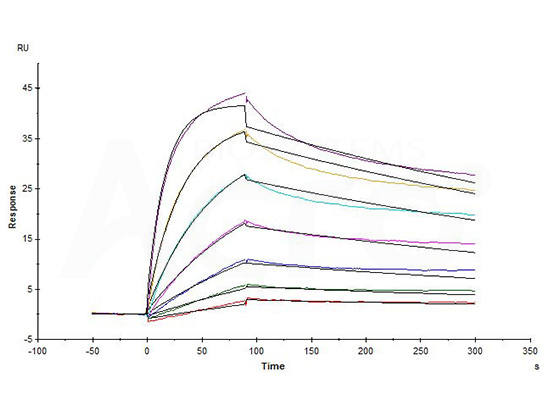
Biotinylated Human CD20 Full Length, His,Avitag (Cat. No. CD0-H82E5) captured on Biotin CAP-Series S Sensor Chip can bind Rituximab with an affinity constant of 1.73 nM as determined in a SPR assay (in presence of DDM and CHS) (Biacore T200) (QC tested).
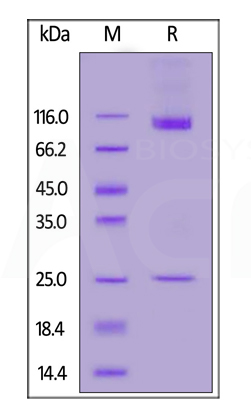
Human CD133 Full Length, His Tag (Nanodisc) (Cat. No. CD3-H52H1) on SDS-PAGE under reducing (R) condition. The gel was stained overnight with Coomassie Blue. The purity of the protein is greater than 90%.

Immobilized Human CD133 Full Length Protein, His Tag (Cat. No. CD3-H52H1) at 1 μg/mL (100 μL/well) can bind Monoclonal Anti-Human CD133 Antibody, Human IgG1 with a linear range of 0.2-4 ng/mL (QC tested).

Authors: Lionel Rougé et al.
Journal: Science
Authors: Paige M. Glumac , Aaron M. LeBeau.
Journal: Glumac and LeBeau Clin Trans Med
Authors: Kenji Maeda, Debananda Das et al.
Journal: Journal of Biological Chemistry
Authors: Line Barington, Pia C. Rummel, et al.
Journal: Journal of Biological Chemistry
This web search service is supported by Google Inc.
