 Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit! Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit!
 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
| Cat. No. | Species | Product Description | Structure | Purity | Feature |
|---|---|---|---|---|---|
| IT1-H82W1 | Human | Biotinylated Human Integrin alpha 4 beta 1 (ITGA4&ITGB1) Heterodimer Protein, His,Avitag™&Tag Free |  |

|

|
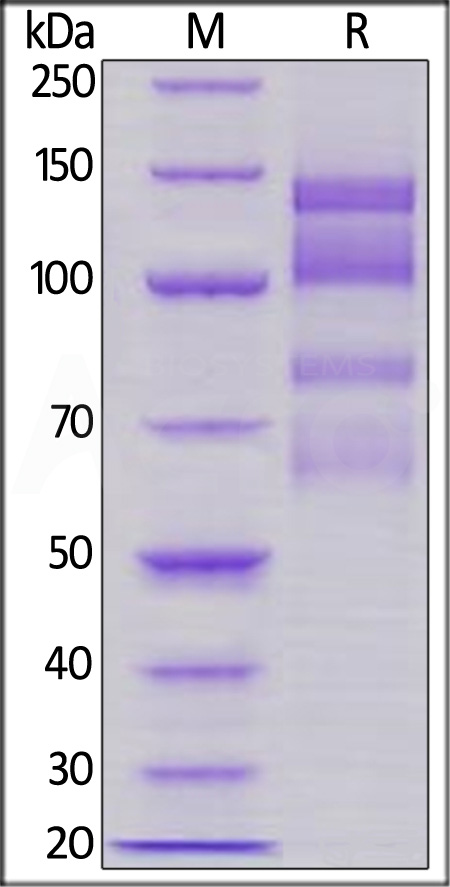
| IT1-H52W1 | Human | Human Integrin alpha 4 beta 1 (ITGA4&ITGB1) Heterodimer Protein, His Tag&Tag Free |  |
|
|

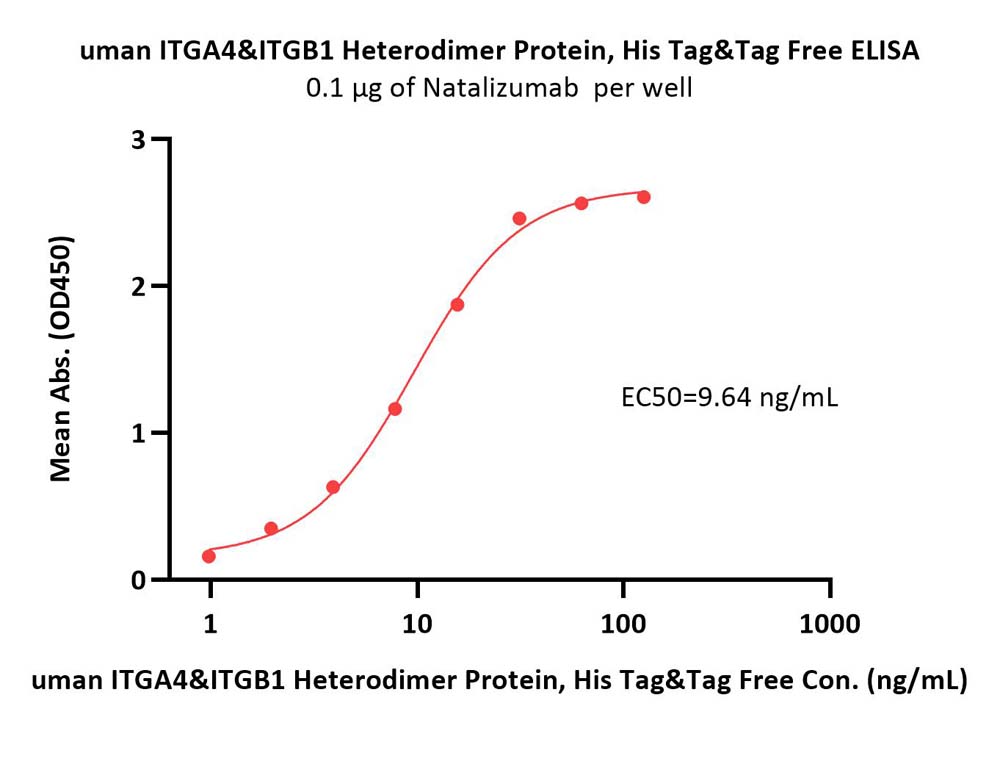
Immobilized Natalizumab at 1 μg/mL (100 μL/well) can bind Human ITGA4&ITGB1 Heterodimer Protein, His Tag&Tag Free (Cat. No. IT1-H52W1) with a linear range of 1-16 ng/mL (QC tested).
| Name | Research Code | Research Phase | Company | First Brand Name | First Approved Country | First Indication | First Approved Company | First Approved Date | Indications | Clinical Trials |
|---|---|---|---|---|---|---|---|---|---|---|
| Carotegrast methyl | AJM-300 | Approved | Ea Pharma, Eisai Co Ltd | Carogra | Japan | Colitis, Ulcerative | Ea Pharma Co Ltd | 2022-03-28 | Colitis, Ulcerative | Details |
| Natalizumab | BG-0002; TY-21.6; AN-10022; BG-00002; AN-100226; BG-0002-E | Approved | Biogen Inc, Perrigo Llc | Tysabri, Antegran, Antegren | Japan | Multiple Sclerosis | Biogen Inc | 2004-11-23 | Multiple Sclerosis, Relapsing-Remitting; Epilepsies, Partial; Arthritis, Rheumatoid; Graft vs Host Disease; Stroke; Multiple Sclerosis, Chronic Progressive; Multiple Sclerosis; Multiple Myeloma; Demyelinating Diseases; Myositis, Inclusion Body; Crohn Disease | Details |
| Name | Research Code | Research Phase | Company | Indications | Clinical Trials |
|---|---|---|---|---|---|
| RBx-7796 | RBx-7796 | Clinical | Sun Pharmaceutical Industries Ltd | Rhinitis, Allergic; Asthma | Details |
| ATL-1102 | ATL-1102; TV-1102; ATL/TV-1102; ISIS-107248 | Phase 2 Clinical | Ionis Pharmaceuticals Inc | Multiple Sclerosis; Muscular Dystrophy, Duchenne | Details |
| 7HP-349 | 7HP-349; 7-HP-349 | Details | |||
| Firategrast | T-0047; SB-683699 | Mitsubishi Tanabe Pharma | Details | ||
| Natalizumab biosimilar (Polpharma Biologics) | PB-006 | Phase 3 Clinical | Polpharma Biologics Sa | Multiple Sclerosis, Relapsing-Remitting | Details |
| LLP2A alendronate | Phase 1 Clinical | University Of California | Osteonecrosis; Bone Diseases, Metabolic; Osteoporosis; Bone Diseases | Details |
This web search service is supported by Google Inc.
