 Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit! Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit!
 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
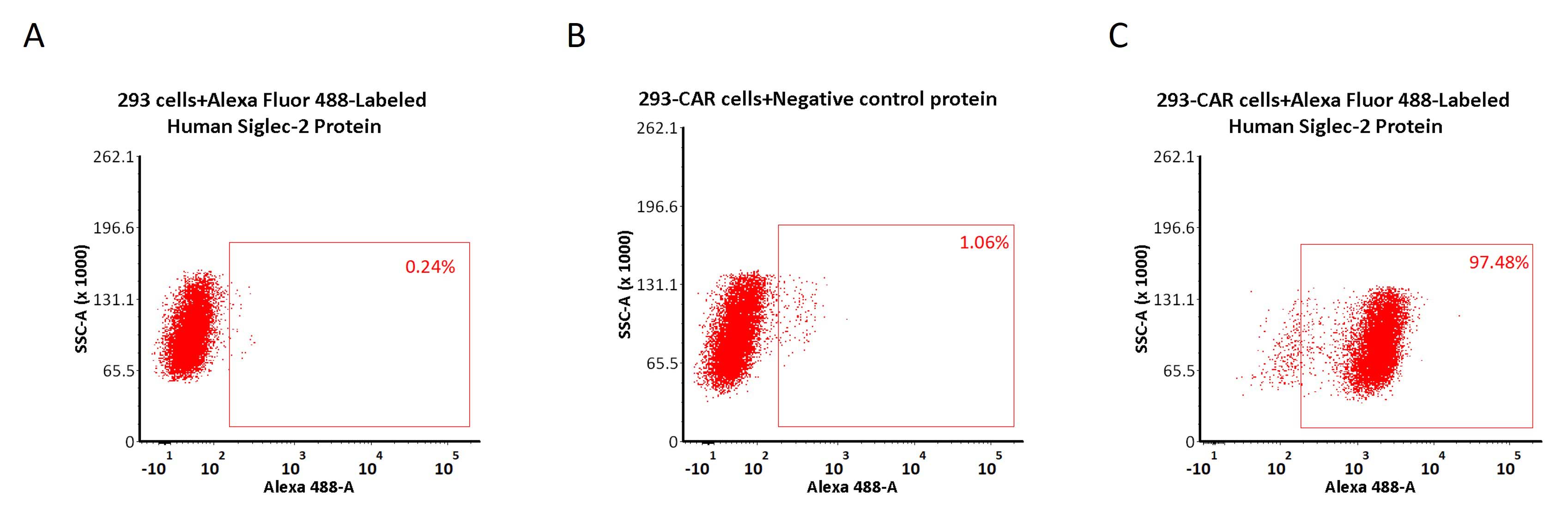
5e5 of anti-CD22 CAR-293 cells were stained with 100 μL of 10 μg/mL of Alexa Fluor 488-Labeled Human Siglec-2, His Tag (Cat. No. SI2-HA2H7) and negative control protein respectively (Fig. C and B), and non-transfected 293 cells were used as a control (Fig. A). Alexa 488 signal was used to evaluate the binding activity (QC tested).
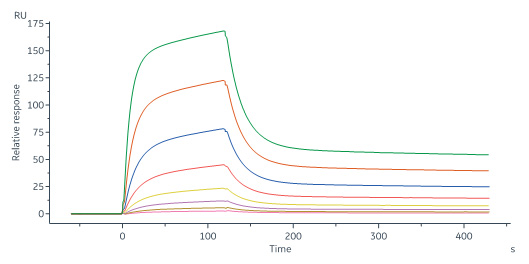
Monoclonal Anti-Human Siglec-2 Antibody, Human IgG1 captured on CM5 chip via Anti-human IgG Fc antibodies surface can bind Human Siglec-2 (176-687), His Tag (Cat. No. SI2-H52H8) with an affinity constant of 0.121 μM as determined in a SPR assay (Biacore 8K) (Routinely tested).
| Name | Research Code | Research Phase | Company | First Brand Name | First Approved Country | First Indication | First Approved Company | First Approved Date | Indications | Clinical Trials |
|---|---|---|---|---|---|---|---|---|---|---|
| Moxetumomab pasudotox | HA-22; GCR-8015; CAT-8015 | Approved | Astrazeneca Plc | Lumoxiti | United States | Leukemia, Hairy Cell | Innate Pharma | 2018-09-13 | Leukemia; Leukemia, Hairy Cell; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma; Lymphoma, Non-Hodgkin; Leukemia, Prolymphocytic; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Inotuzumab Ozogamicin | WAY-207294; PF-5208773; CMC-544 | Approved | Pfizer Inc, Ucb | Besponsa | Mainland China | Precursor B-Cell Lymphoblastic Leukemia-Lymphoma | Pfizer Investment Co Ltd | 2017-06-28 | Lymphoma, B-Cell; Hematologic Neoplasms; Leukemia, Myelogenous, Chronic, BCR-ABL Positive; Leukemia; Lymphoma, Large B-Cell, Diffuse; Blast Crisis; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Follicular; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Precursor T-Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell; Lymphoma; Lymphoma, Non-Hodgkin; Burkitt Lymphoma | Details |
| Name | Research Code | Research Phase | Company | Indications | Clinical Trials |
|---|---|---|---|---|---|
| RD-102 | RD-102 | Phase 1 Clinical | Leukemia, Lymphoid; Lymphoma, Large B-Cell, Diffuse | Details | |
| Anti-CD22 CAR T cell therapy (Shanghai GeneChem) | Phase 1 Clinical | Shanghai Genechem Co Ltd | Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details | |
| Anti-CD19 and anti-CD22 CAR T cell therapy (Seattle Children's Hospital) | Phase 1 Clinical | Seattle Children'S Hospital | Precursor Cell Lymphoblastic Leukemia-Lymphoma | Details | |
| Anti-CD22 CAR-T cell therapy (No. 307 Hospital) | Phase 2 Clinical | The Fifth Medical Center Of Pla General Hospital (Formerly 307 Hospital) | Precursor Cell Lymphoblastic Leukemia-Lymphoma | Details | |
| anti-CD22-CAR-transduced T cells(Southwest Hospital) | Phase 2 Clinical | Southwest Hospital | Leukemia; Lymphoma | Details | |
| Anti-CD22 CAR T cell therapy (Wuhan Bio-Raid) | Phase 1 Clinical | Wuhan Bio-Raid Biotechnology | Lymphoma, B-Cell; Multiple Myeloma | Details | |
| Anti-CD19/22-CAR vector-transduced T cell therapy (Chinese PLA General Hospital) | Phase 2 Clinical | People'S Liberation Army General Hospital Military Service | Leukemia; Lymphoma | Details | |
| CTA-101 | CTA-101 | Phase 1 Clinical | Nanjing Bioheng Biotech Co Ltd, Nanjing Medical University, Xuzhou Medical University (Xzmu) | Precursor B-Cell Lymphoblastic Leukemia-Lymphoma | Details |
| CD19/CD22 dual-target CAR-T cell therapy (Shenzhen University General Hospital) | Phase 2 Clinical | Shenzhen University General Hospital | Precursor Cell Lymphoblastic Leukemia-Lymphoma | Details | |
| Anti-CD19 and anti-CD22 CAR-T cell therapy(IASO Biotherapeutics) | CT-120 | Phase 2 Clinical | Nanjing Iaso Biotherapeutics Co Ltd | Lymphoma, B-Cell; Leukemia, B-Cell; Lymphoma, Non-Hodgkin | Details |
| CD19x22 CAR T Cell Therapy (University Of Colorado Denver) | Phase 1 Clinical | University Of Colorado, Denver, Usa | Lymphoma, Non-Hodgkin | Details | |
| IMJ-995 | IMJ-995 | Novartis Pharma Ag | Details | ||
| CD22 CAR-T Cell Therapy (Hebei Senlang Biotechnology) | Hebei Senlang Biological Technology Co Ltd | Details | |||
| CD22-targeted CAR-T cell therapy (PersonGen Biomedicine) | Persongen Biotherapeutics | Details | |||
| BAY-1862864 | BAY-1862864 | Bayer AG | Details | ||
| Anti-CD22-CAR T cell therapy (Kecellitics Biotech) | Kecellitics Biotech Company Ltd | Details | |||
| Suciraslimab | SM-06; SM-03 | Phase 3 Clinical | Sinomab Bioscience Ltd | Arthritis, Rheumatoid; Lupus Erythematosus, Systemic; Lymphoma, Non-Hodgkin | Details |
| TRPH-222 | CD22-4AP; TRPH-222; CAT-02-106 | Catalent Inc | Details | ||
| bispecific CD19/CD22 CAR T-cell therapy (University of Colorado) | Phase 1 Clinical | University Of Colorado | Lymphoma, B-Cell; Lymphoma, Non-Hodgkin | Details | |
| CART22-cells | CART22-cells; JJO686 | Phase 1 Clinical | Novartis Pharma Ag, University Of Pennsylvania | Lymphoma, B-Cell; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell | Details |
| CD19/CD22 dual-target CAR-T (Yake biotechnology) | Phase 2 Clinical | Shanghai YaKe Biotechnology Co Ltd | Precursor B-Cell Lymphoblastic Leukemia-Lymphoma | Details | |
| Anti-CD22/CD19 monoclonal antibody-toxin conjugate | Phase 1 Clinical | The University Of Texas Southwestern Medical Center | Lymphoma, B-Cell; Precursor Cell Lymphoblastic Leukemia-Lymphoma | Details | |
| CD19-CD22 CAR-T cells (Shanghai Ultra-T Immune Therapeutics) | Phase 2 Clinical | Shanghai Ultra-T Immune Therapeutics Co LTD | Lymphoma, B-Cell; Multiple Myeloma; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell | Details | |
| Bispecific CD19/22 CAR T cells | Phase 2 Clinical | Lymphoma, Non-Hodgkin; Lymphoma | Details | ||
| Anti-CD19 anti-CD22 bispecific chimeric antigen receptor T cell therapy (Hrain Biotechnology) | Phase 1 Clinical | Hrain Biotechnology Co Ltd | Leukemia, Lymphoid | Details | |
| CAR-20-19-22 | CAR-20-19-22 | Phase 1 Clinical | The Medical College Of Wisconsin Nonprofit | Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| CD19/22 Bi-specific CAR-T Cell Therapy(Shenzhen Geno-Immune Medical Institute) | 4SCAR19/22 | Phase 2 Clinical | Shenzhen Geno-Immune Medical Institute | Lymphoma, B-Cell; Leukemia, B-Cell | Details |
| 4SCAR-T cell therapy (Shenzhen Geno-Immune Medical Institute) | 4SCAR-T | Phase 2 Clinical | Shenzhen Geno-Immune Medical Institute | Hematologic Neoplasms; Autoimmune Diseases; Neuroblastoma | Details |
| Chimeric antigen receptor T cell therapeutics (targeted CD19/CD20/CD22/CD30,Shanghai Unicar-Therapy Bio-medicine) | Phase 2 Clinical | Shanghai Unicar-Therapy Bio-Medicine Technology Co Ltd | Pancreatic Neoplasms; Lymphoma, Non-Hodgkin | Details | |
| JCAR-018 | JCAR-018 | Phase 1 Clinical | National Cancer Institute, Opus Bio | Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Follicular; Lymphoma, Non-Hodgkin | Details |
| CD20/CD22 dual Targeted CAR T-cell therapy (Zhejiang University) | Phase 1 Clinical | Zhejiang University, Shanghai YaKe Biotechnology Co Ltd | Hematologic Neoplasms | Details | |
| Anti-CD19/CD22 bispecific CAR-T cell therapy (The Second Affiliated Hospital of Henan University of Traditional Chinese Medicine) | Phase 1 Clinical | The Second Affiliated Hospital Of Henan University Of Traditional Chinese Medicine | Precursor T-Cell Lymphoblastic Leukemia-Lymphoma | Details | |
| Dual Anti-CD22/CD19 Chimeric Antigen Receptor-directed T Cells (CART2219.1) therapy (KK Women's and Children's Hospital) | Phase 2 Clinical | Kk Women'S And Children'S Hospital | Leukemia, Lymphoid; Precursor Cell Lymphoblastic Leukemia-Lymphoma | Details | |
| UCART-22 | UCART-22 | Phase 1 Clinical | Cellectis Sa | Precursor B-Cell Lymphoblastic Leukemia-Lymphoma | Details |
| Dual anti-CD19/CD22 CAR‐T cell therapy (B-cell malignancies, Stanford University) | Phase 1 Clinical | Stanford University, Orca Biosystems Inc | Leukemia, Myelogenous, Chronic, BCR-ABL Positive; Neoplasm, Residual; Leukemia, Lymphoid; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma | Details | |
| GC-022 | GC-022; GC-022F | Phase 2 Clinical | Gracell Biotechnologies (Shanghai) Co Ltd | Hematologic Neoplasms; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Non-Hodgkin | Details |
| Anti-CD19/CD22 CAR-T cell therapy (Jiao Tong University) | Phase 1 Clinical | Shanghai Jiaotong University, Shanghai General Hospital | Precursor Cell Lymphoblastic Leukemia-Lymphoma | Details | |
| ThisCAR-T-22(Fundamenta Therapeutics) | ThisCAR-T-22 | Phase 1 Clinical | Fundamenta Therapeutics Ltd | Lymphoma, B-Cell | Details |
| Anti CD22 CAR-T cell therapy (Shanghai Yake Biotechnology) | Phase 2 Clinical | Shanghai YaKe Biotechnology Co Ltd, The First Affiliated Hospital Of Zhejiang University School Of Medicine, Beijing Gao Boren Hospital Co Ltd, Chengdu USino Technology Biology Co Ltd | Lymphoma, B-Cell; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma | Details | |
| JNJ-75348780 | Phase 1 Clinical | Janssen Research & Development Llc | Lymphoma, B-Cell; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details | |
| Anti-CD19 and anti-CD22 CAR T cell therapy (Hebei Senlang Biotechnology) | Phase 1 Clinical | Hebei Senlang Biological Technology Co Ltd | Leukemia, B-Cell | Details | |
| CD19/CD20 Dual CAR-T Cell Therapy (China Immunotech) | HX-s001; YTS101; HX-s001/YTS101; HXYT-001 | Phase 1 Clinical | Beijing Qingyi Taike Pharmaceutical Technology Co Ltd, China Immunotech Co Ltd | Lymphoma, B-Cell; Lymphoma, Large B-Cell, Diffuse; Leukemia, B-Cell; Lymphoma, Non-Hodgkin | Details |
| CD19- and CD22 specific CAR (Seattle Children's Hospital) | Phase 1 Clinical | Seattle Children'S Hospital | Leukemia; Lymphoma | Details | |
| UCART-20x22 | UCART-20x22 | Phase 2 Clinical | Cellectis Sa | Lymphoma, Non-Hodgkin | Details |
| CD19/CD22 CAR-T cell therapy (Federal Research Institute of Pediatric Hematology, Oncology and Immunology) | Phase 2 Clinical | Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Burkitt Lymphoma | Details | ||
| Autologous CD19/CD22 chimeric antigen receptor T-cell therapy (MD Anderson Cancer Center) | Phase 2 Clinical | The University Of Texas MD Anderson Cancer Center | Neoplasm, Residual; Leukemia, Lymphoid; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details | |
| Dual specificity CD19 and CD20 or CD22 CAR-T cell therapy(Chinese PLA General Hospital) | Phase 2 Clinical | Pla General Hospital | Lymphoma, B-Cell; Leukemia, B-Cell | Details | |
| SCRI-CAR22v2 | Phase 2 Clinical | Seattle Children'S Hospital | Leukemia; Lymphoma | Details | |
| Epratuzumab-cys-tesirine | ADCT-602 | Phase 2 Clinical | Adc Therapeutics Sa | Leukemia, Myelogenous, Chronic, BCR-ABL Positive; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell | Details |
This web search service is supported by Google Inc.
