 Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit! Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit!
 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
| Cat. No. | Species | Product Description | Structure | Purity | Feature |
|---|---|---|---|---|---|
| IGR-M5253 | Mouse | Mouse IGF-I R / CD221 Protein, Fc Tag |  |
|

|
| CNIH-ATP102 | Human | NIH-3T3/Human IGF-1 R Stable Cell Line Development Service | |||
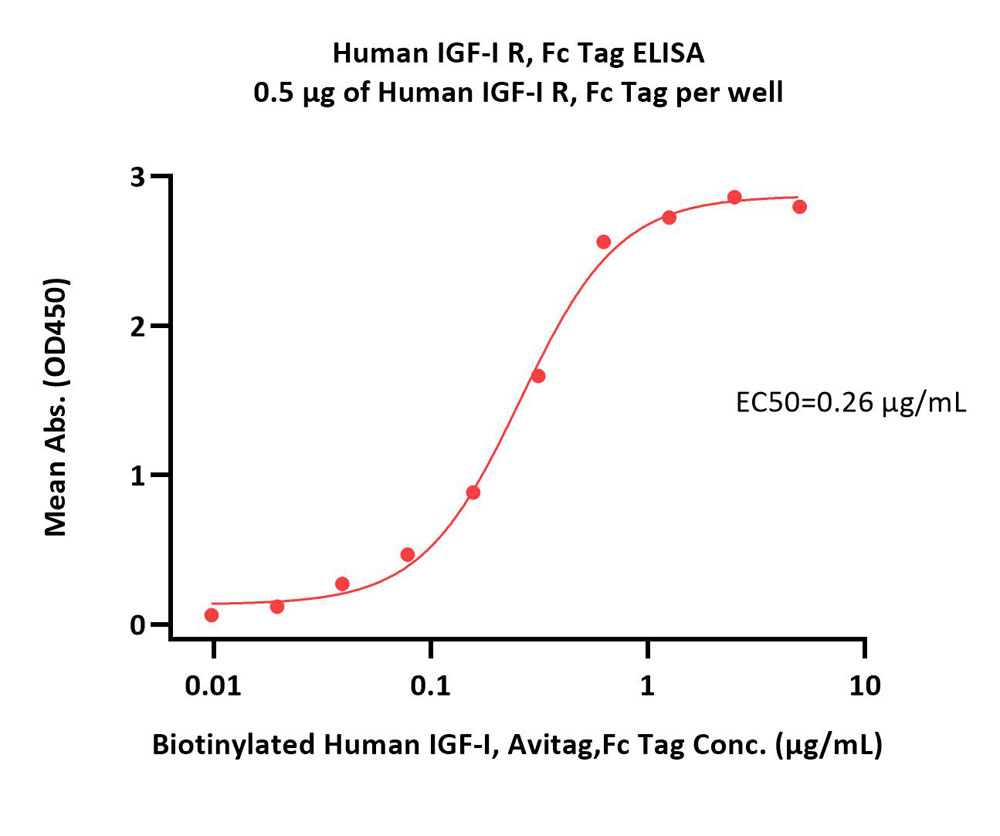
| IGR-H5253 | Human | Human IGF-I R / CD221 Protein, Fc Tag |  |
|
|
| IGR-H82E3 | Human | Biotinylated Human IGF-I R / CD221 Protein, His,Avitag™ |  |
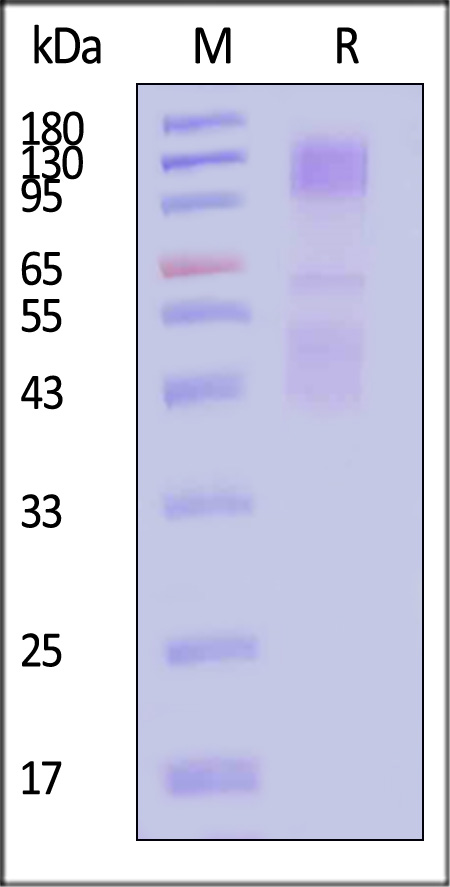
|

|
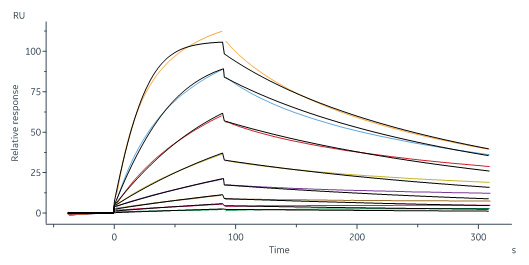
| IGR-H5229 | Human | Human IGF-I R / CD221 Protein, His Tag (MALS verified) |  |

|


|
| IGR-C5225 | Cynomolgus | Cynomolgus IGF-I R / CD221 Protein, His Tag (MALS verified) |  |

|
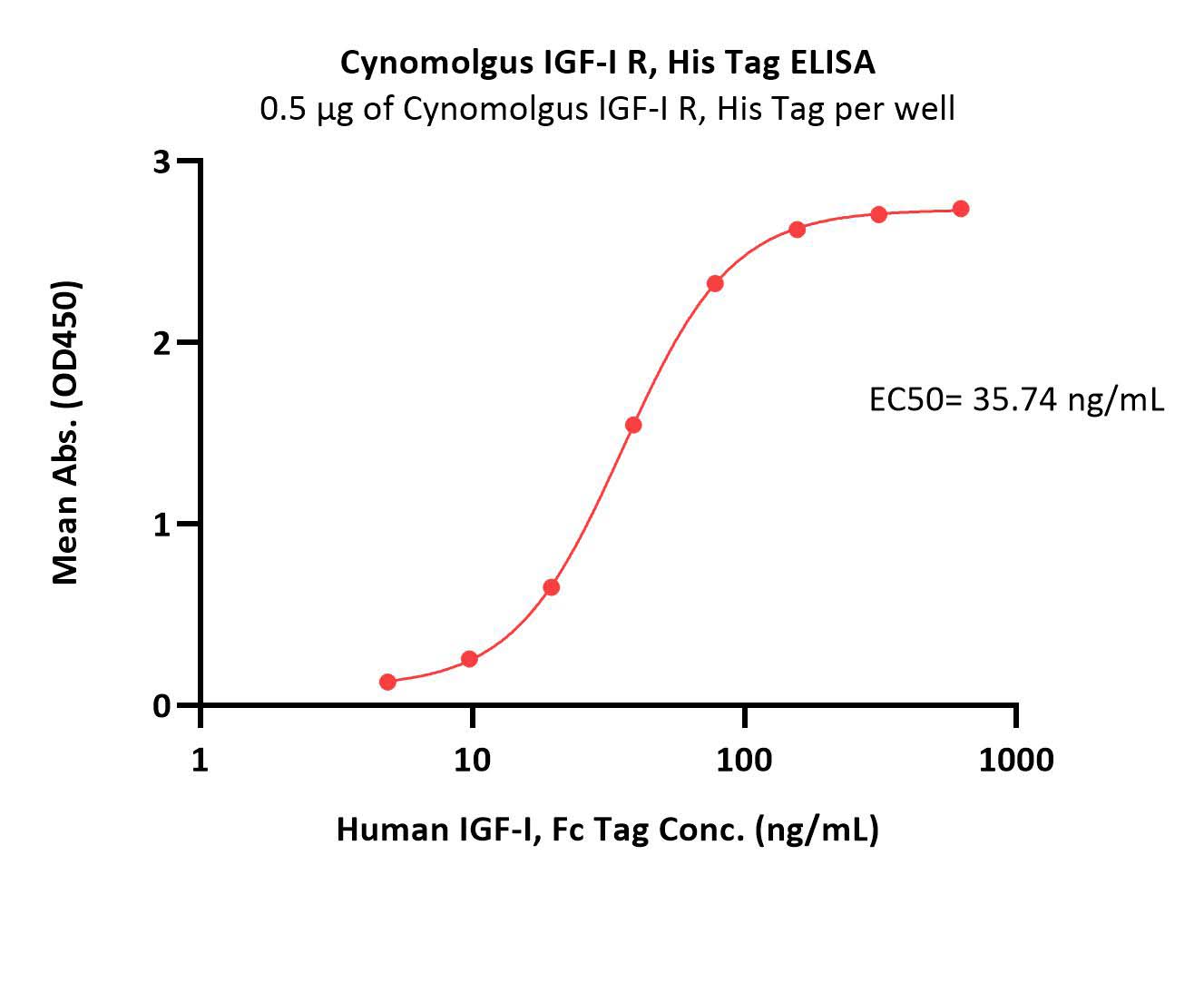
|
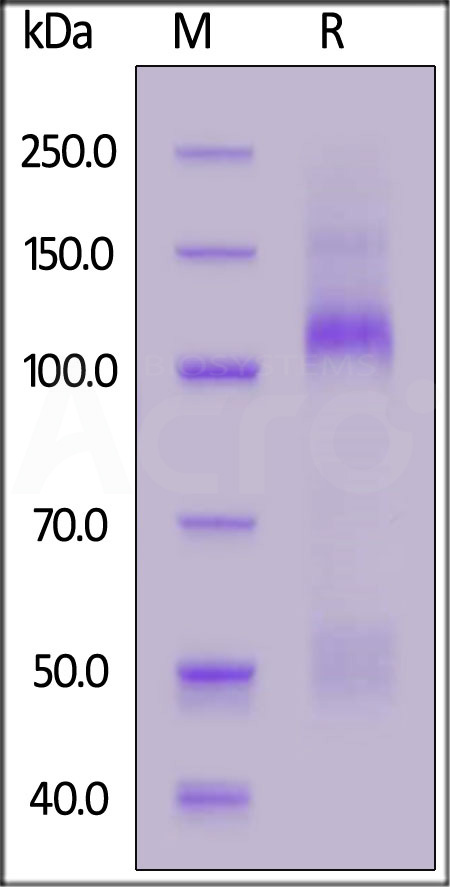
| IGR-R5224 | Rat | Rat IGF-I R / CD221 Protein, His Tag (MALS verified) |  |

|
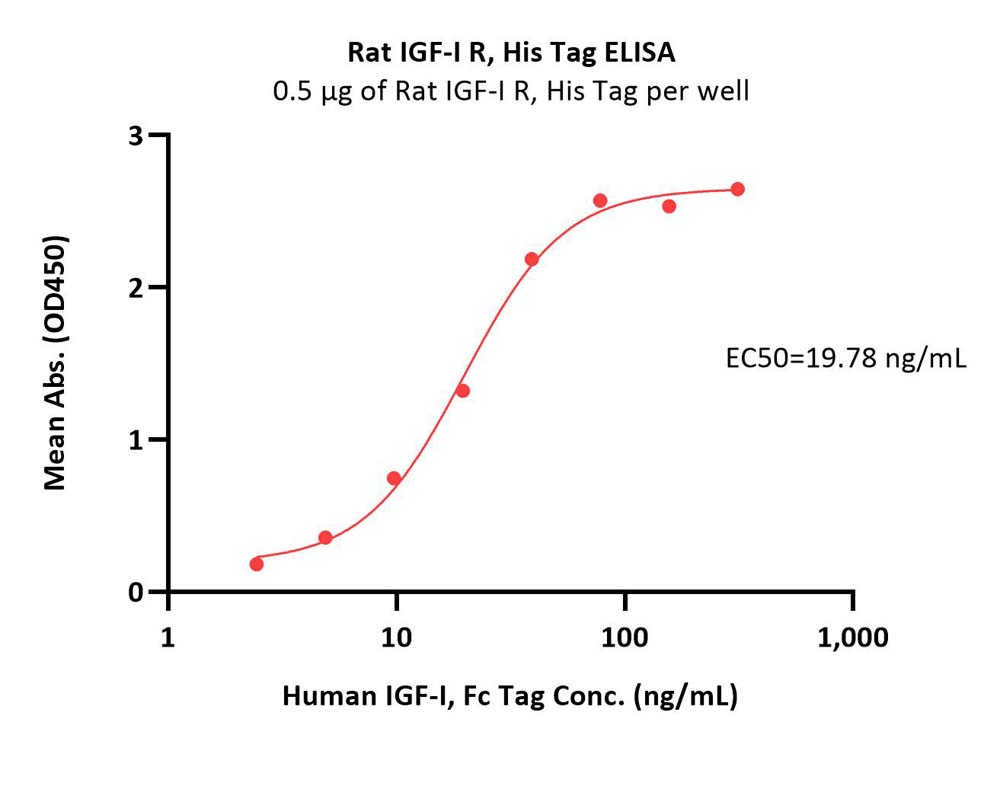
|
| IGR-M5223 | Mouse | Mouse IGF-I R / CD221 Protein, His Tag |  |
|
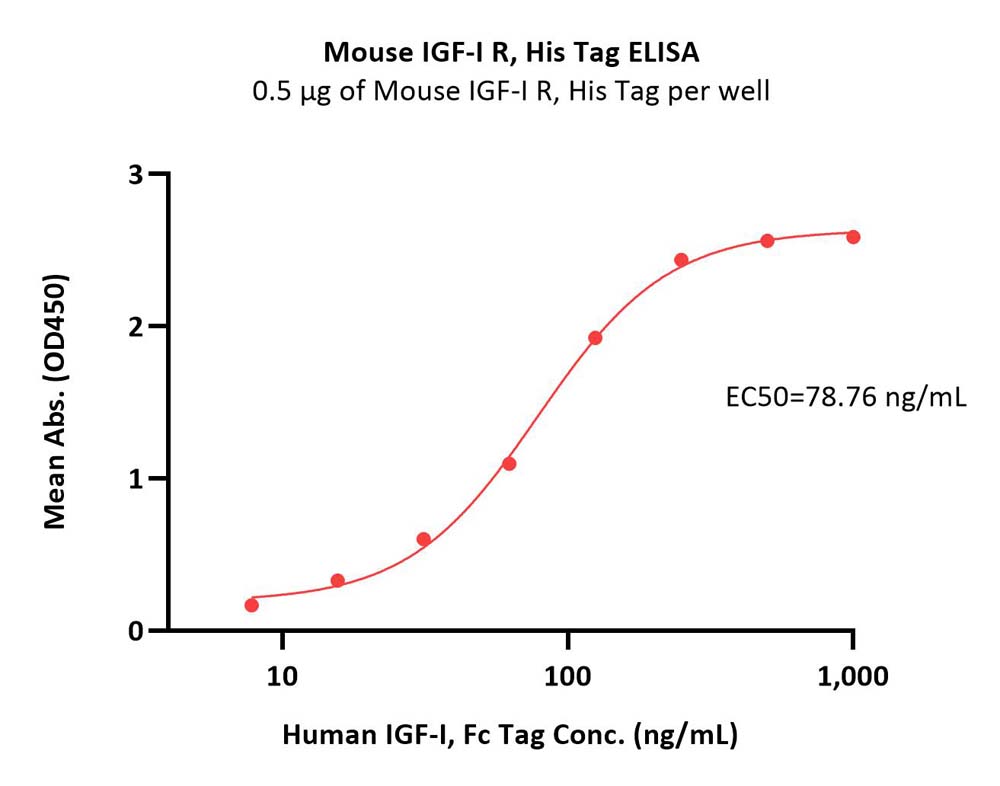
|
| Name | Research Code | Research Phase | Company | First Brand Name | First Approved Country | First Indication | First Approved Company | First Approved Date | Indications | Clinical Trials |
|---|---|---|---|---|---|---|---|---|---|---|
| IGF-1 (Biogen/Pharmacia & Upjohn) | Approved | Biogen Inc, Pharmacia & Upjohn | Igef | Sweden | Dwarfism | null | 1994-01-01 | Dwarfism | Details | |
| Mecasermin (Astellas/OrphanPacific) | Approved | Astellas Pharma Inc | Somazon | Japan | Dwarfism; Diabetes Mellitus, Lipoatrophic; Growth hormone deficiency | null | 1994-10-05 | HIV Infections; Diabetes Mellitus, Lipoatrophic; Heart Failure; Myocardial Infarction; Multiple Sclerosis; Lipodystrophy; Dwarfism; Growth hormone deficiency | Details | |
| Mecasermin (Ipsen) | IGF-1; FK-780; MKN-031 | Approved | Ipsen | Increlex | EU | Laron Syndrome | Ipsen Pharma | 2005-08-30 | Failure to Thrive; Autism Spectrum Disorder; X-Linked Combined Immunodeficiency Diseases; Phelan-McDermid syndrome; Growth Disorders; Muscular Dystrophy, Duchenne; Laron Syndrome | Details |
| Brigatinib | AP-26113 | Approved | Ariad, Takeda Pharmaceutical Co Ltd | Alunbrig | Mainland China | Carcinoma, Non-Small-Cell Lung | Takeda (China) International Trading Co Ltd | 2017-04-28 | Solid tumours; Ependymoma; Carcinoma; Neoplasms; Neurofibromatosis 2; Myofibroma; Lymphoma, Large-Cell, Anaplastic; Granuloma, Plasma Cell; Lung Neoplasms; Brain metastases; Carcinoma, Non-Small-Cell Lung; Sarcoma, Kaposi; Meningioma; Neurilemmoma; Neuroma, Acoustic | Details |
| Teprotumumab | R-1507; RG-1507; RO-4858696; HZN-001 | Approved | Genmab A/S, F. Hoffmann-La Roche Ltd | Tepezza | United States | Graves Ophthalmopathy | Horizon Therapeutics Ireland | 2020-01-21 | Neoplasms; Graves Ophthalmopathy; Sarcoma; Breast Neoplasms; Diabetic macular oedema; Scleroderma, Diffuse; Carcinoma, Non-Small-Cell Lung | Details |
| Ceritinib | LDK-378; NVP-LDK378; NVP-LDK378-NX | Approved | Novartis Pharma Ag | 赞可达, Zykadia | Mainland China | Carcinoma, Non-Small-Cell Lung | Novartis Europharm Ltd | 2014-04-29 | Hepatic Insufficiency; Melanoma; Adenocarcinoma; Carcinoma, Non-Small-Cell Lung; Thyroid Neoplasms; Brain metastases; Esophageal adenocarcinoma; Granuloma, Plasma Cell; Lymphoma, Large-Cell, Anaplastic; Hematologic Neoplasms; Colorectal Neoplasms; Cholangiocarcinoma; Thyroid Carcinoma, Anaplastic; Neoplasms; Pancreatic Neoplasms; Glioblastoma; Stomach Neoplasms | Details |
| Name | Research Code | Research Phase | Company | Indications | Clinical Trials |
|---|---|---|---|---|---|
| 124I-CPD-1023-figitumumab | 124I-CPD-1028; 125I-CPD-1028; CPD-1028-[124I]; CPD-1028-[125I]; FPX-1028 | Phase 1 Clinical | Fusion Pharma | Neoplasms | Details |
| Insulin-Like Growth Factor 1 (Wright State Physicians) | Phase 1 Clinical | Wright State Physicians | Nasolabial fold wrinkles; Sunburn | Details | |
| Lonigutamab | VB-421 | Phase 1 Clinical | Pierre Fabre Group | Graves Ophthalmopathy | Details |
| IGF-1 receptor oligodeoxynucleotide-based immunotherapy (Thomas Jefferson University) | Phase 1 Clinical | Sidney Kimmel Cancer Center | Glioma | Details | |
| BVS-857 | BVS-857 | Novartis Pharma Ag | Details | ||
| VPI-2690B | VPI-2690B | University Of North Carolina At Chapel Hill | Details | ||
| KW-2450 | KW-2450 | Kyowa Hakko Kirin Co Ltd | Details | ||
| BIIB-022 | BIIB-022 | Biogen Inc | Details | ||
| Mecasermin Rinfabate | OHB-607; SHP-607; HGT-ROP-001; rhIGF-I/rhIGFBP-3; TAK-607 | Phase 2 Clinical | Insmed | Noonan Syndrome; Failure to Thrive; Cerebral Intraventricular Hemorrhage; Myotonic Dystrophy; Bronchopulmonary Dysplasia; Retinopathy of Prematurity; Muscular Dystrophies; Laron Syndrome | Details |
| Teprotumumab biosimilar(Innovent) | IBI-311 | Phase 1 Clinical | Innovent Biologics(Suzhou) Co Ltd | Graves Ophthalmopathy | Details |
| Picropodophyllin | PPP; AXL-1717; NSC-36407 | Phase 2 Clinical | Axelar Ab | Astrocytoma; Carcinoma, Non-Small-Cell Lung | Details |
| Linsitinib | OSI-906; ASP-7487; OSI-906AA | Phase 3 Clinical | Astellas Pharma Inc, National Cancer Institute | Endocrine System Diseases; Paraganglioma; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Carcinoma, Squamous Cell; Eye Diseases; Orbital Diseases; Gastrointestinal Stromal Tumors; Colorectal Neoplasms; Breast Neoplasms; Prostatic Neoplasms; Sarcoma, Ewing; Graves Ophthalmopathy; Solid tumours; Multiple Myeloma; Adrenocortical Carcinoma; Small Cell Lung Carcinoma; Pancreatic Neoplasms; Skin Neoplasms; Neoplasms; Hashimoto Disease; Carney Complex; Liver Neoplasms; Chondrosarcoma; Thyroid Diseases; Ovarian Neoplasms | Details |
| Cixutumumab | A-12; IMC-A12; LY-3012217; NSC-742460 | Phase 2 Clinical | Eli Lilly And Company | Osteosarcoma; Gliosarcoma; Adenocarcinoma, Bronchiolo-Alveolar; Colorectal Neoplasms; Medullary thyroid cancer (MTC); Neuroblastoma; Gastrinoma; Neurofibrosarcoma; Retinoblastoma; Brain Stem Neoplasms; Breast Neoplasms; Prostatic Neoplasms; Sarcoma; Sarcoma, Ewing; Lymphoma, Mantle-Cell; Liposarcoma; Glucagonoma; Esophageal adenocarcinoma; Neuroectodermal Tumors, Primitive, Peripheral; Adenocarcinoma; Paraganglioma; Melanoma; Breast Neoplasms, Male; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Sarcoma, Alveolar Soft Part; Adrenocortical Carcinoma; Glioma; Rhabdomyosarcoma, Alveolar; Esophageal Squamous Cell Carcinoma; Hemangiopericytoma; Carcinoma, Neuroendocrine; Lung Neoplasms; Pinealoma; Squamous Cell Carcinoma of Head and Neck; Carcinoma, Merkel Cell; Insulinoma; Hemangiosarcoma; Mesenchymoma; Hepatoblastoma; Stomach Neoplasms; Esophageal Neoplasms; Carcinoma; Desmoplastic Small Round Cell Tumor; Kidney Neoplasms; Fibrosarcoma; Chondrosarcoma; Liver Neoplasms; Head and Neck Neoplasms; Leiomy | Details |
| Ganitumab | AMG-479 | Phase 3 Clinical | Amgen Inc | Prostatic Neoplasms; Neuroectodermal Tumors, Primitive, Peripheral; Gastrointestinal Neoplasms; Neoplasm Metastasis; Carcinoma, Non-Small-Cell Lung; Rhabdomyosarcoma, Alveolar; Lymphoma; Lung Neoplasms; Neuroectodermal Tumors, Primitive; Colorectal Neoplasms; Sarcoma, Ewing; Breast Neoplasms; Sarcoma; Solid tumours; Pancreatic Neoplasms; Colonic Neoplasms; Small Cell Lung Carcinoma; Neoplasms; Rhabdomyosarcoma, Embryonal; Rectal Neoplasms; Carcinoid Tumor; Ovarian Neoplasms; Rhabdomyosarcoma; Bone metastases; Intestinal Neoplasms | Details |
| Humanized IgG1κ monoclonal antibody(Viridian) | ZB-001; AVE-1642; VRDN-001 | Phase 2 Clinical | Viridian Therapeutics Inc | Solid tumours; Multiple Myeloma; Graves Ophthalmopathy; Breast Neoplasms | Details |
| FPI-1434 | [225Ac]-FPI-1434; FPX-01 | Phase 2 Clinical | Fusion Pharma | Ovarian Neoplasms; Solid tumours; Squamous Cell Carcinoma of Head and Neck; Triple Negative Breast Neoplasms; Neoplasms; Uveal melanoma; Adrenocortical Carcinoma; Breast Neoplasms; Endometrial Neoplasms; Uterine Cervical Neoplasms | Details |
| PHP-1003 | PHP-1003; PHP1003 | Phase 1 Clinical | Graves Ophthalmopathy | Details | |
| lonigutamab ugodotin | W-0101 | Phase 2 Clinical | Pierre Fabre Group | Solid tumours | Details |
| Conteltinib | CT-707; SY-707 | Phase 3 Clinical | Shouyao Holding (Beijing) Co Ltd, Centaurus Biopharma Co Ltd | Pancreatic Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| VRDN-002 | VRDN-002 | Phase 1 Clinical | Viridian Therapeutics Inc | Graves Ophthalmopathy | Details |
| CT-102 | CT-102 | Phase 2 Clinical | Hangzhou Tianlong Pharmaceutical Co Ltd, Institute Of Radiation And Radiation Medicine, Chinese Academy Of Military Medical Sciences | Liver Neoplasms; Carcinoma, Hepatocellular | Details |
This web search service is supported by Google Inc.
