 Order Online! Now! Get your $50 coupon for online order.
Order Online! Now! Get your $50 coupon for online order. Order Online! Now! Get your $50 coupon for online order.
Order Online! Now! Get your $50 coupon for online order.
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
 Fill out organ-on-a-chip questionnaire to win a FREE gift!
Fill out organ-on-a-chip questionnaire to win a FREE gift!  Fill out organ-on-a-chip questionnaire to win a FREE gift!
Fill out organ-on-a-chip questionnaire to win a FREE gift!
> Chemokine family proteins 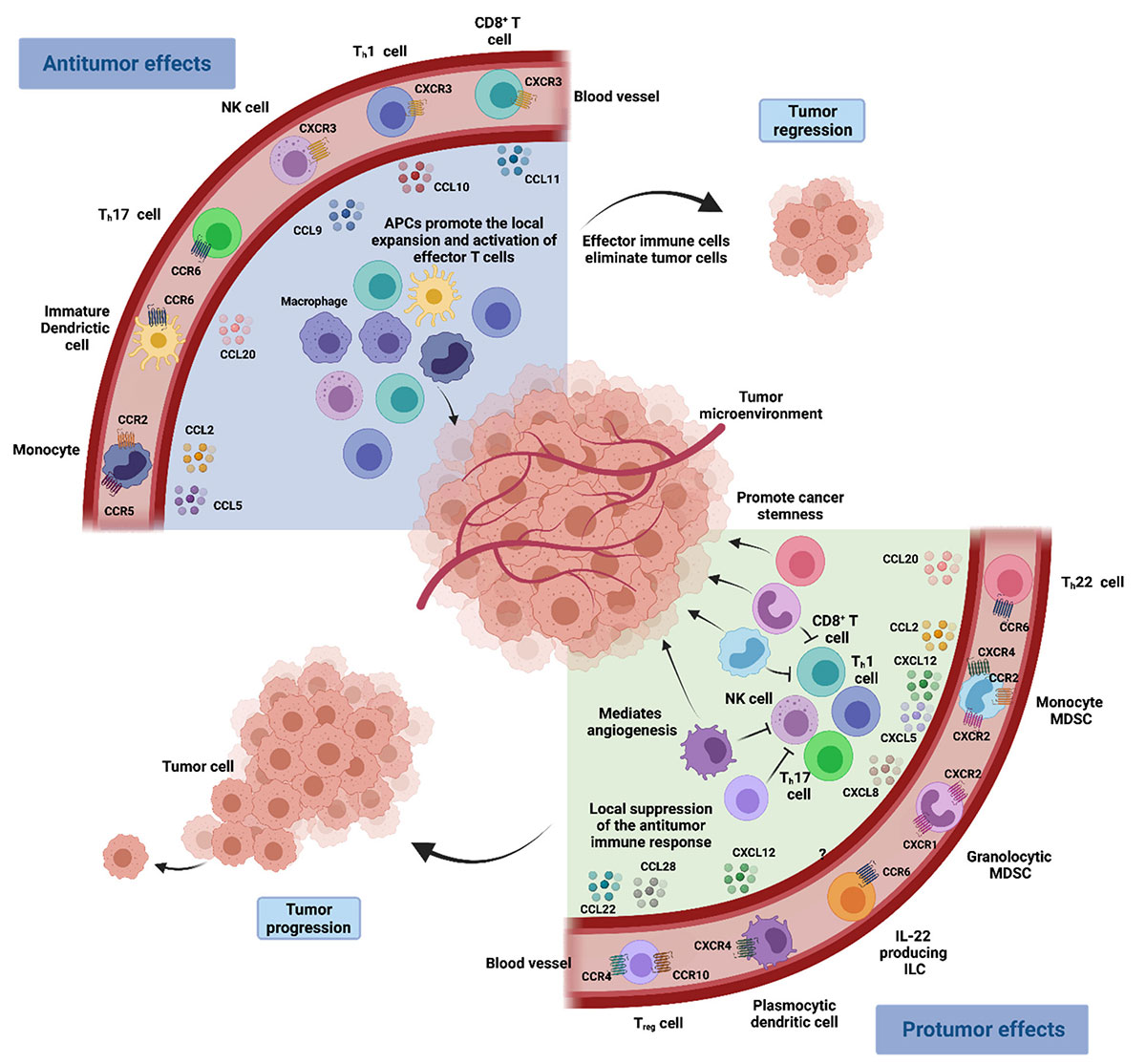
Chemokine network in the tumoral microenvironment immune response
Chemokines are small cytokines or signaling proteins secreted by cells. According to the number and arrangement of conserved L-Cysteine (C) at the N-terminal, chemokines can be classified into four classes: C, CC, CXC and CX3C. These chemokines signal through the seven-pass transmembrane chemokine receptor of G-protein coupling on the cell surface, which stimulate the cell directional migration. Homeostatic chemokines are constitutively expressed under physiological conditions and play a role in cell migration and homing, whereas inflammatory chemokines are rapidly secreted at sites of inflammation, thereby recruiting effector cells into inflamed tissues. Thus, chemokines play a central role in the development and homeostasis of the immune system and are involved in promoting the activation, differentiation, proliferation and apoptosis of immune cells and in all protective or destructive immune and inflammatory responses.
Furthermore, chemokine signaling and the chemotaxis of various cell populations also play a central role in the tumor microenvironment (TME). In the TME, many chemokines are released by immune cells, tumor cells and tumor-associated cells. These chemokines can change with time and space, attracting different types of tumor-promoting and anti-tumor immune cells. The migration of immune cells will further affect the progression and metastasis of tumors, and directly affect the immune response. Studies have revealed that chemokines have pleiotropic functions that affect anti-tumor immune responses and tumor progression. With this in mind, therapeutic strategies that utilize or target chemokines have been established over the past decade, leading to the testing of many new drug candidates in clinical trials. Notably, in chemokine-based therapies, chemokines are highly redundant in binding to receptors, the same receptor can bind to different chemokines, and vice versa. Therefore, only by simultaneously inhibiting chemokine and its receptor with the same physiological function can the therapeutic effect be healthier and lasts longer.
ACROBiosystems has developed a series of CC, CXC and CX3C subfamily chemokine proteins expressed by HEK293. The products are available in a variety of species and labels. In addition, based on the “FLAG” technology platform, we developed full-length chemokine receptor such as CCR5, CCR8, CXCR3, CXCR4, CXCR5, CXCR6, etc. which with high biological activity was verified by binding to antibody. Our recombinant chemokine family proteins can accelerate your scientific research and drug development programs.
![]() Various molecules, species, and tags
Various molecules, species, and tags
![]() High purity verified by SDS-PAGE
High purity verified by SDS-PAGE
![]() High structural homogeneity verified by SEC-MALS and DLS
High structural homogeneity verified by SEC-MALS and DLS
![]() High activity verified by ELISA/SPR
High activity verified by ELISA/SPR
![]() Highly stable and convenient for storage and transportation
Highly stable and convenient for storage and transportation
| Molecule | Cat. No. | Species | Product Description | Preorder/Order |
|---|---|---|---|---|
| CCL1 | CC1-H82E9 | Human | Biotinylated Human CCL1 / I-309 / C-C motif chemokine 1 Protein, His,Avitag™ (MALS verified) | |
| CC1-H5254 | Human | Human CCL1 / I-309 Protein, Fc Tag (MALS verified) | ||
| CCL2 | CC2-H5255 | Human | Human CCL2 / MCP-1 Protein, Fc Tag (MALS verified) | |
| CCL3 | CC3-H5249 | Human | Human CCL3 / MIP-1 alpha Protein, His Tag | |
| CCL5 | CC5-H5251 | Human | Human CCL5/RANTES Protein, Fc Tag (MALS verified) | |
| CCL7 | CC7-H52H6 | Human | Human CCL7 / MCP-3 Protein, His Tag | |
| CCL14 | CC4-H5249 | Human | Human CCL14 / HCC-1 Protein, His Tag | |
| CCL17 | CC7-H5256 | Human | Human CCL17 / TARC Protein, Fc Tag (MALS verified) | |
| CCL22 | CC2-H5257 | Human | Human CCL22 / MDC Protein, Fc Tag | |
| CCL24 | CC4-H52H1 | Human | Human CCL24/Eotaxin-2 Protein, His Tag | |
| CXCL1 | CX1-H5253 | Human | Human CXCL1 Protein, Fc Tag | |
| CXCL4 | CX4-H52H9 | Human | Human CXCL4 / PF4 Protein, His Tag | |
| CXCL10 | CX0-H51H1 | Human | Human CXCL10 / IP-10 Protein, His Tag | |
| CXCL13 | CX3-H1249 | Human | Human CXCL13 / BCA-1 Protein, His Tag | |
| CXCL16 | CX6-H5254 | Human | Human CXCL16 Protein, Fc Tag | |
| CX3CL1 | CX1-H5221 | Human | Human CX3CL1 / Fractalkine Protein, His Tag | |
| CX1-H8221 | Human | Biotinylated Human CX3CL1 Protein, His Tag, ultra sensitivity (primary amine labeling) | ||
| CX1-C5222 | Cynomolgus | Cynomolgus CX3CL1 / Fractalkine Protein, His Tag | ||
| CXCR3 | CX3-H52P4 | Human | Human CXCR3 Full Length Protein-VLP | |
| CX3-H52D5 | Human | Human CXCR3 Protein, Flag,His Tag (Detergent) | ||
| CX3-M52P7 | Mouse | Mouse CXCR3 Full Length Protein (VLP) | ||
| CXCR4 | CX4-H5219 | Human | Human CXCR4 / CD184 Full Length Protein-VLP | |
| CX4-H5269 | Human | Human CXCR4 / CD184 Protein, Fc Tag | ||
| CXCR5 | CX5-H52P4 | Human | Human CXCR5 Full Length Protein-VLP | |
| CX5-H52D6 | Human | Human CXCR5 Protein, Flag,His Tag (Detergent) | ||
| CXCR6 | CX6-H52P3 | Human | Human CXCR6 / CD186 Full Length Protein (VLP) | |
| CCR4 | CC4-H52P3 | Human | Human CCR4 Full Length Protein (VLP) | |
| CCR5 | CC5-H52P3 | Human | Human CCR5 Full Length Protein-VLP | |
| CC5-H52P5 | Human | Fluorescent Human CCR5 Full Length Protein (VLP) | ||
| CC5-H52D1 | Human | Human CCR5 Protein, Flag,His Tag (Detergent) | ||
| CCR6 | CC6-H52P3 | Human | Human CCR6 Full Length Protein (VLP) | |
| CCR7 | CC7-H52P3 | Human | Human CCR7 Full Length Protein (VLP) | |
| CCR8 | CC8-H52P4 | Human | Human CCR8 Full Length Protein-VLP |
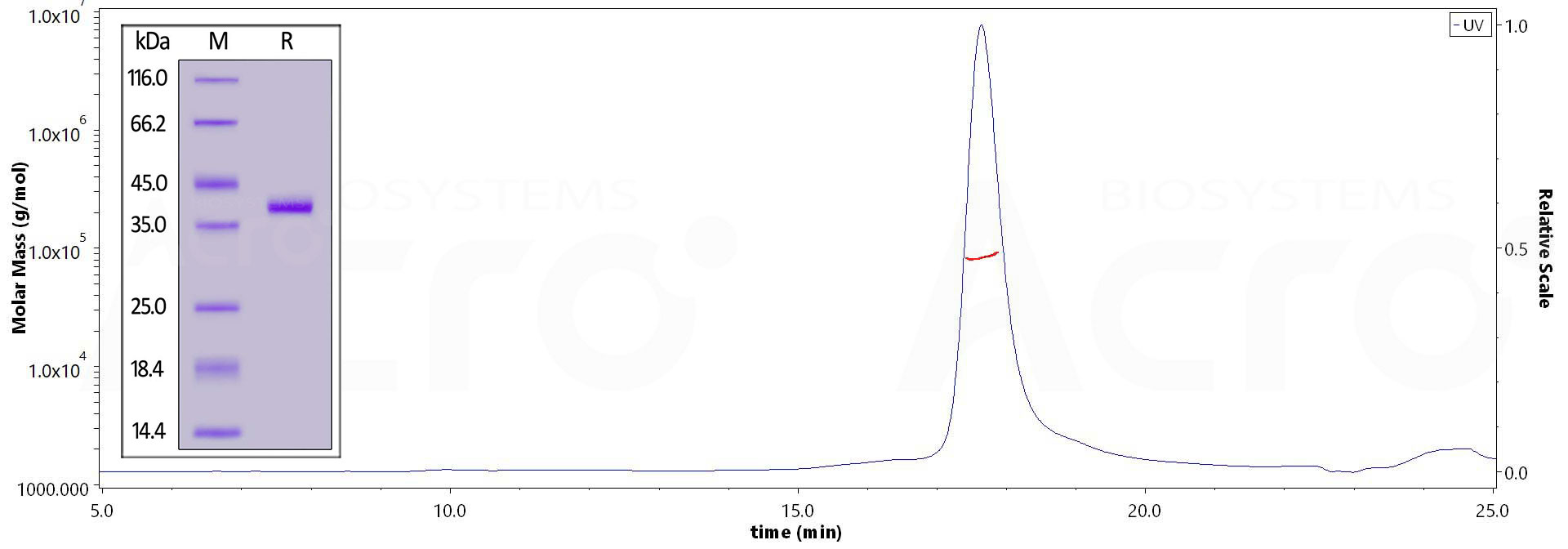
The purity of Human CCL5, Fc Tag (Cat. No. CC5-H5251) is more than 85% and the molecular weight of this protein is around 71-96 kDa verified by SEC-MALS.
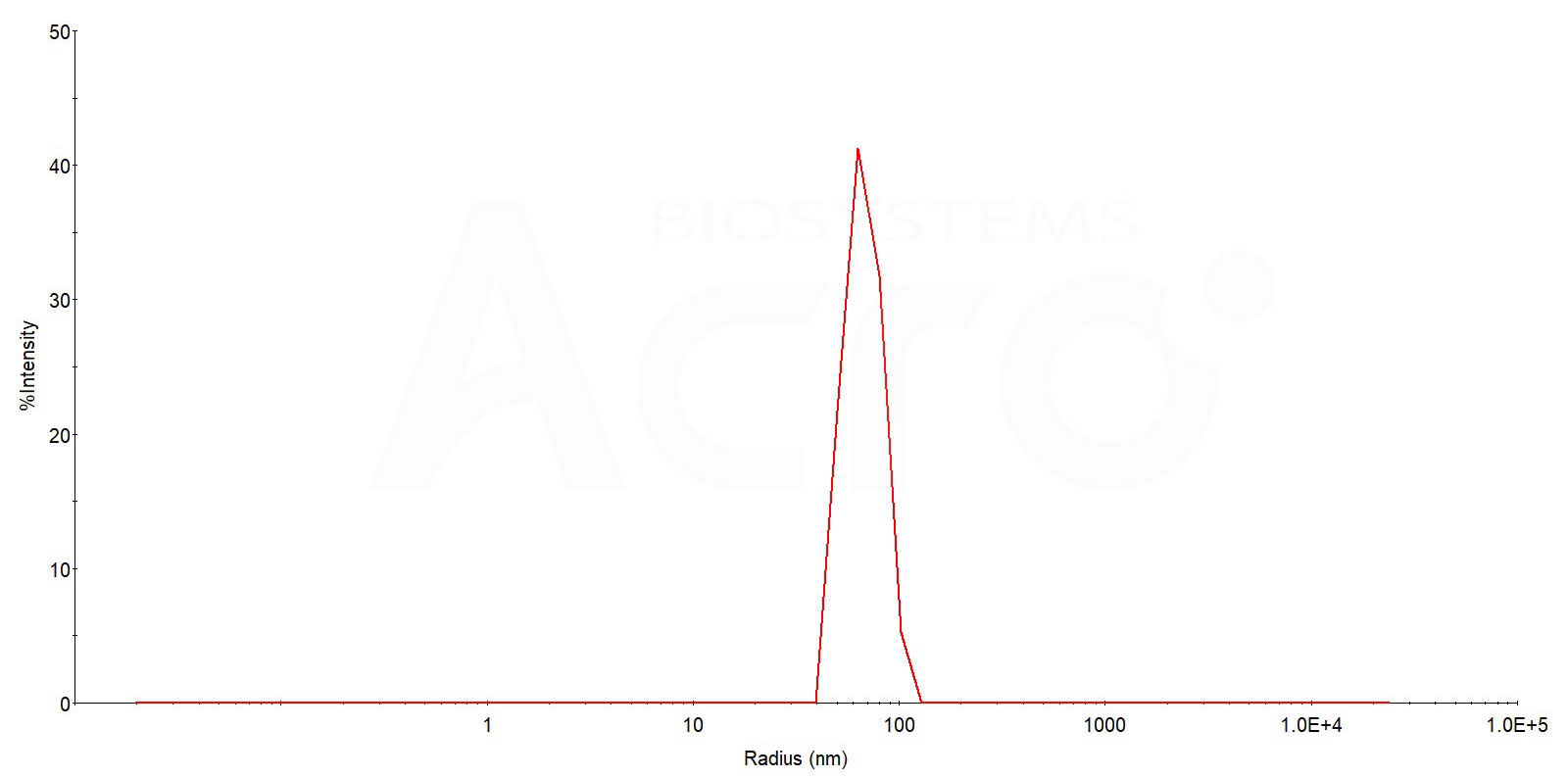
The mean peak Radius of VLP (Cat. No. CX3-H52P4) is 60-80 nm with more than 95% intensity as determined by dynamic light scattering (DLS).
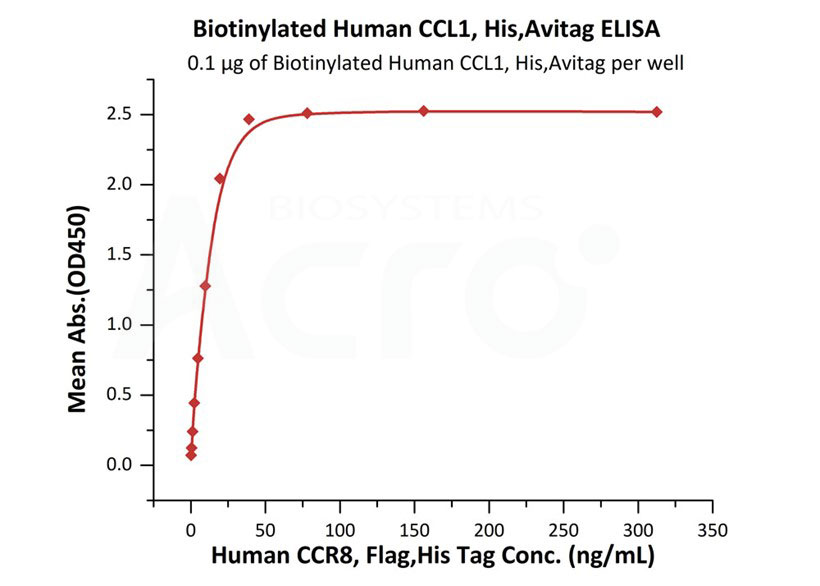
Immobilized Biotinylated Human CCL1, His,Avitag (Cat. No. CC1-H82E9) at 1 μg/mL (100 μL/well) on streptavidin (Cat. No. STN-N5116) precoated (0.5 μg/well) plate can bind Human CCR8, Flag,His Tag with a linear range of 0.3-39 ng/mL (QC tested).
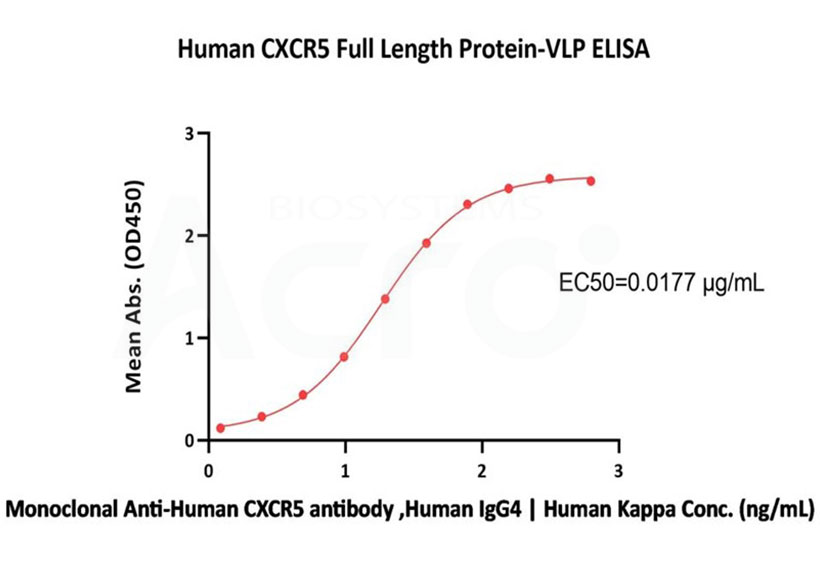
Immobilized Human CXCR5 Full Length Protein-VLP (Cat. No. CX5-H52P4) at 5 μg/mL (100 μL/well) can bind Monoclonal Anti-Human CXCR5 antibody, Human IgG4 | Human Kappa with a linear range of 1-78 ng/mL (QC tested).
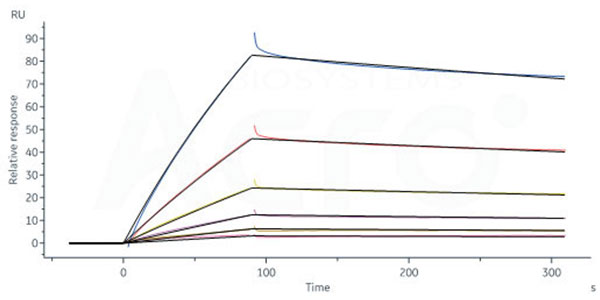
Anti-CXCR3 antibody immobilized on CM5 Chip can bind Human CXCR3 Full Length Protein-VLP (Cat. No. CX3-H52P4) with an affinity constant of 60.9 nM as determined in a SPR assay (Biacore 8K) (Routinely tested).
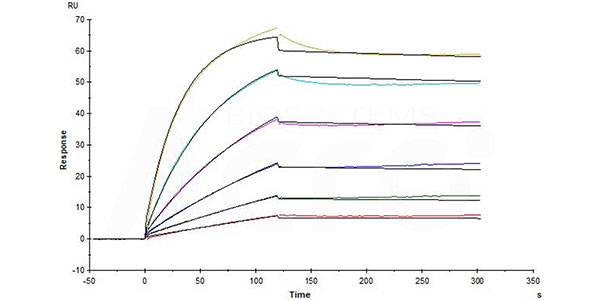
Human CCR5, Flag,His Tag (Cat. No. CC5-H52D1) captured on CM5 chip via anti-His antibody can bind Anti-CCR5 antibody (Human IgG1) with an affinity constant of 0.0737 nM as determined in a SPR assay (in presence of DDM and CHS) (Biacore T200).
This web search service is supported by Google Inc.
